Atom
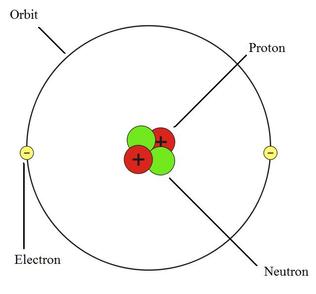
The picture on the left is a helium atom. An atom is the smallest particle of an element, in this case helium, that can exist by itself. A helium atom has 2 protons, 2 electrons and 2 neutrons.
The protons and neutrons are located in the nucleus of the atom. The electrons rotate the nucleus on pathways called atomic orbitals, which are also known as electron shells or energy levels.
Neutron - no charge
Proton - positive charge +
Electron - negative charge -
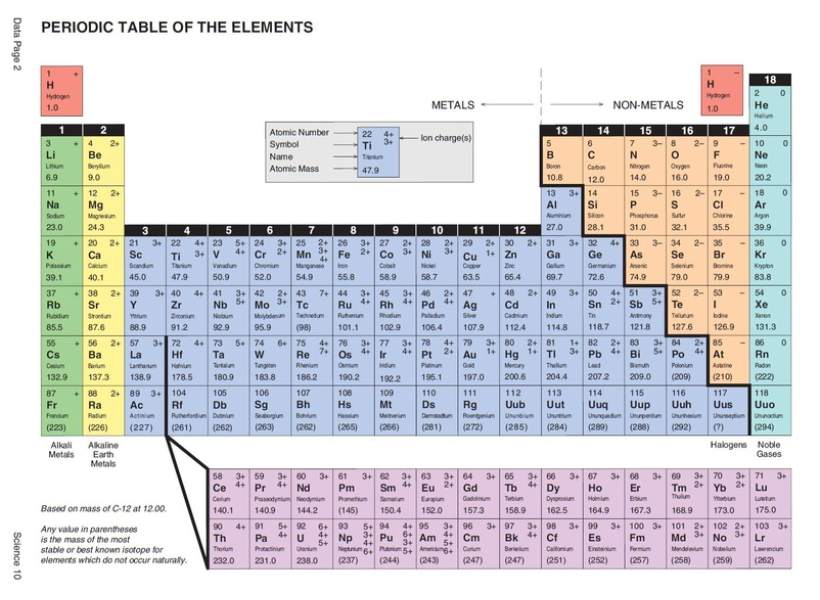
The non-metal elements are found on the right side of the periodic table and the metal elements are found on the left side of the table. Non-metal elements include nitrogen, oxygen, fluorine, neon, phosphorus, silicon etc. Metal elements include sodium, magnesium, iron, cobalt, aluminium etc.
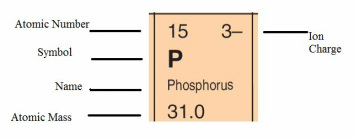
Atomic number = number of electrons
Atomic mass = protons + neutrons
Ion charge is the charge on the atom after it has gained (-) or lost (+) electrons to achieve a stable octet configuration - eight electrons on the outer shell. The first shell of all atoms only contains 2 electrons and all other orbitals contain 8 electrons at a stable state. All atoms strive to achieve a stable configuration. In this case phosphorus has 15 electrons in total. The first 2 electrons go into the first orbital, the next 8 go into the next orbital, and then the remaining 5 electrons go into the outer orbital and they are termed the valence electrons. The P atom needs to gain 3 more electrons to achieve a stable octet (8) configuration. After the phosphorus atom has gained electrons (remember electrons are negative) it becomes an ion with a negative charge of 3.
Atomic mass = protons + neutrons
Ion charge is the charge on the atom after it has gained (-) or lost (+) electrons to achieve a stable octet configuration - eight electrons on the outer shell. The first shell of all atoms only contains 2 electrons and all other orbitals contain 8 electrons at a stable state. All atoms strive to achieve a stable configuration. In this case phosphorus has 15 electrons in total. The first 2 electrons go into the first orbital, the next 8 go into the next orbital, and then the remaining 5 electrons go into the outer orbital and they are termed the valence electrons. The P atom needs to gain 3 more electrons to achieve a stable octet (8) configuration. After the phosphorus atom has gained electrons (remember electrons are negative) it becomes an ion with a negative charge of 3.
Bohr and Lewis Diagrams
A Bohr model of an atom shows the number of neutrons and protons within the nucleus of the atom, as well as all the electrons possessed by the atom. On the other hand, a Lewis diagram is a dot diagram only representing the outermost electron shell's electrons, the valence electrons.

The Bohr Model on the left represents the oxygen atom. Oxygen has 8 electrons in total. Two electrons go into the first orbital or electron shell and the remaining six go into the outermost orbital.

This is a Lewis diagram of oxygen. Only the 6 outer shell electrons, valence electrons, are shown. The 2 electrons that go into the first shell are not shown on a Lewis diagram. The oxygen atom has two unpaired electrons (one electron by itself) and two paired electrons.